Even viability dyes need controls. Don’t have time to “kill” a control for this? Don’t worry- we...
3 Reasons Your Flow Data May be Flawed
Why am I seeing errors in my flow data?
 Flow cytometry is a versatile technology with varied applications throughout cell therapy, pharmaceutical, academic, and clinical institutions. While there are many different ways you can use this technology, the basic principles of panel design, compensation or spectral unmixing, and experimental controls remain the same. However, if these principles are applied incorrectly it can lead to erroneous results leading to setbacks or even worse negative patient outcomes. Let’s explore these three basic principles of flow cytometry and why your flow data may be flawed.
Flow cytometry is a versatile technology with varied applications throughout cell therapy, pharmaceutical, academic, and clinical institutions. While there are many different ways you can use this technology, the basic principles of panel design, compensation or spectral unmixing, and experimental controls remain the same. However, if these principles are applied incorrectly it can lead to erroneous results leading to setbacks or even worse negative patient outcomes. Let’s explore these three basic principles of flow cytometry and why your flow data may be flawed.
Principle 1: Panel Design
The first and most critical step in designing a flow assay begins with panel design. If done improperly, it can lead to issues in resolving populations, which can interfere with the interpretation of your end results. There are several resources to assist you in designing your panels, we recommend starting with this publication and this panel design tool.
To begin the panel design process, you need to organize your antigens into primary, secondary, and tertiary classifications. This first step helps to assign the appropriate fluorophores to the correct antigens by considering their expression pattern. While errors can be made in this first step, to really visualize those errors you need to proceed with the next step of panel design which involves evaluating the impact of spread in your panel.
If you do not evaluate spread in your panel, your data can be flawed since the presence of spread impacts your ability for population resolution. You can empirically predict spread by creating a spillover spread matrix (SSM), first reported here. Not only will a SSM expose potential incompatible fluorophores and spread within your flow panel but it can also show how the panel will act instrument to instrument, which is useful for standardizing instruments with the same panel. A SSM can be made by staining well-expressed markers like CD4 in each fluorophore in your panel. The spillover spread value correlates with the amount of spread between fluors, and is interpreted as a heat map. Areas highlighted in darker red show where spread is relatively high in your panel.
See example:
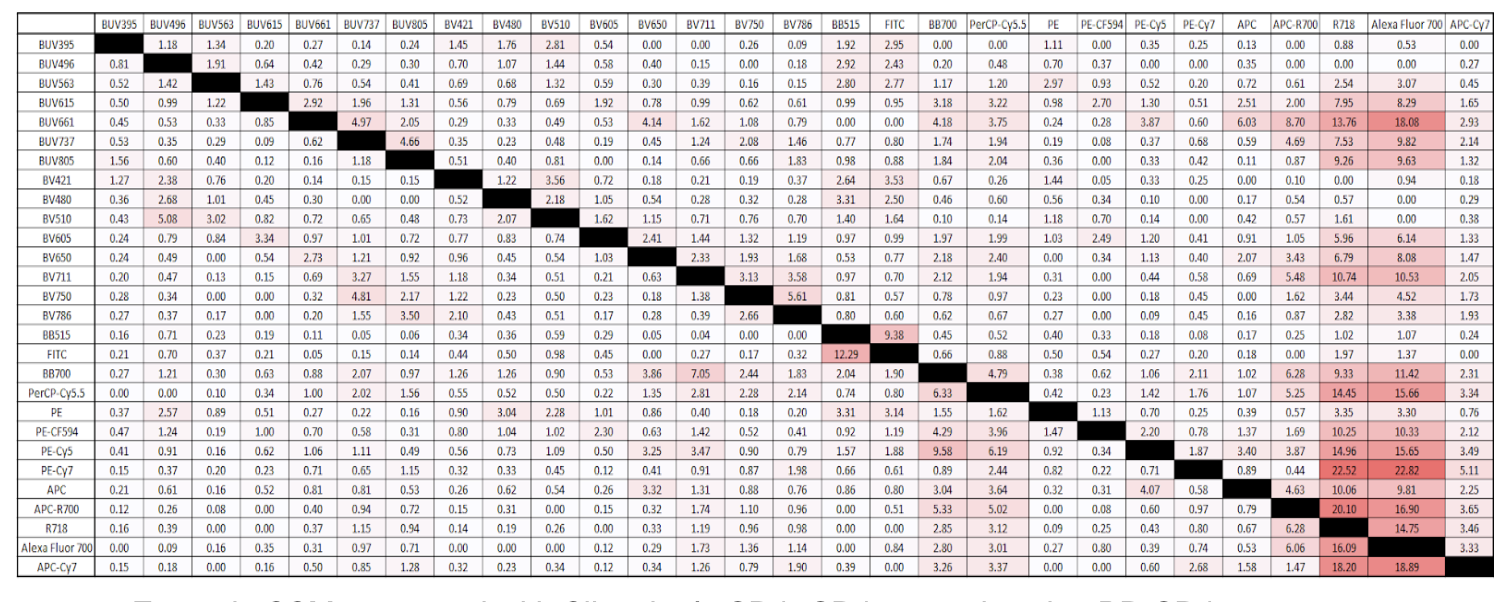 Example SSM generated with Slingshot’s CD4+CD4- controls using BD CD4 Fluorochrome Evaluation Kit
Example SSM generated with Slingshot’s CD4+CD4- controls using BD CD4 Fluorochrome Evaluation Kit
Principle 2: Compensation or Spectral Unmixing with Single Stained Controls
After you have designed your panel, the next basic principle of flow cytometry involves single stained controls for compensation or spectral unmixing.
There are three golden rules you must follow to avoid introducing errors into your flow data.
Rule 1
The first rule is that your single stained control spectrum must match the spectrum of that same fluor in the multicolor. Breaking this first rule can lead to spectral mismatches or compensation errors which equate to inaccurate results. Examples of breaking this rule would be using a different tandem dye in your single stained control than that which is in your multicolor or differences in how you prepare your single stained controls vs the multicolor sample.
Rule 2
The second rule is that your positive and negative particles should be well separated and the autofluorescence of your negative particle must match the autofluorescence of your positive particle. With this rule you want to ensure that first, if you are using single stained cells you have an unstained cell control or vice versa if using a synthetic control material for compensation or unmixing. Also, you want to match the negative population to the cells of interest for the marker you are looking at.
Rule 3
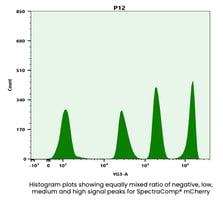
The last rule is that your single stained control should be as bright or brighter than your multicolor sample and as a subrule to this you also need to collect enough events to provide accurate data for the algorithms. This rule often applies to cells which can have different levels of brightness for a given fluor. If you gate on the dimmer population rather than the brightest it can result in compensation or unmixing errors. Adding onto this, too few events can lead to calculation errors in the compensation or unmixing software as well as making it difficult to accurately gate the populations (see example below).
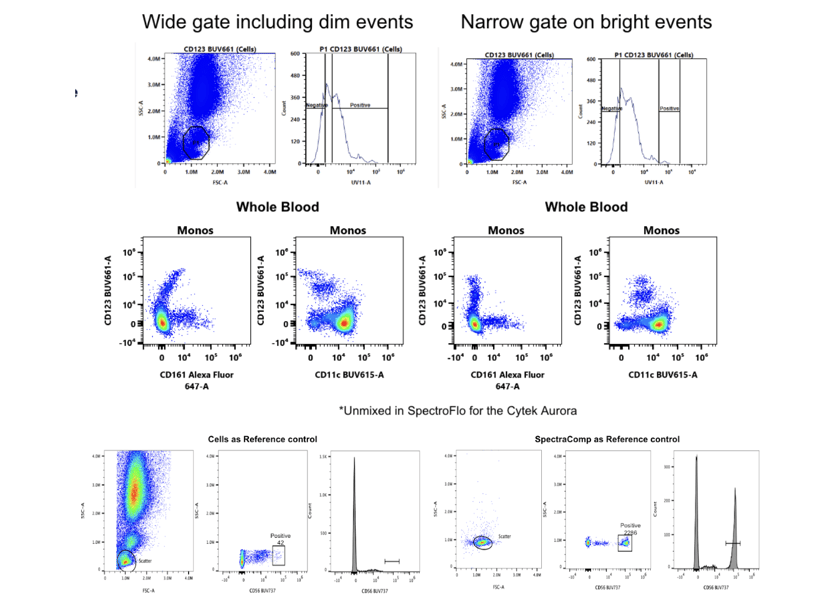
Principle 3: Experimental Controls
The last principle of flow cytometry which helps to ensure accurate results is the use of appropriate experimental controls. We already touched on some of these controls above such as CD4 controls for SSM generation, or single stained controls, however, experimental controls go beyond panel design and compensation or unmixing.
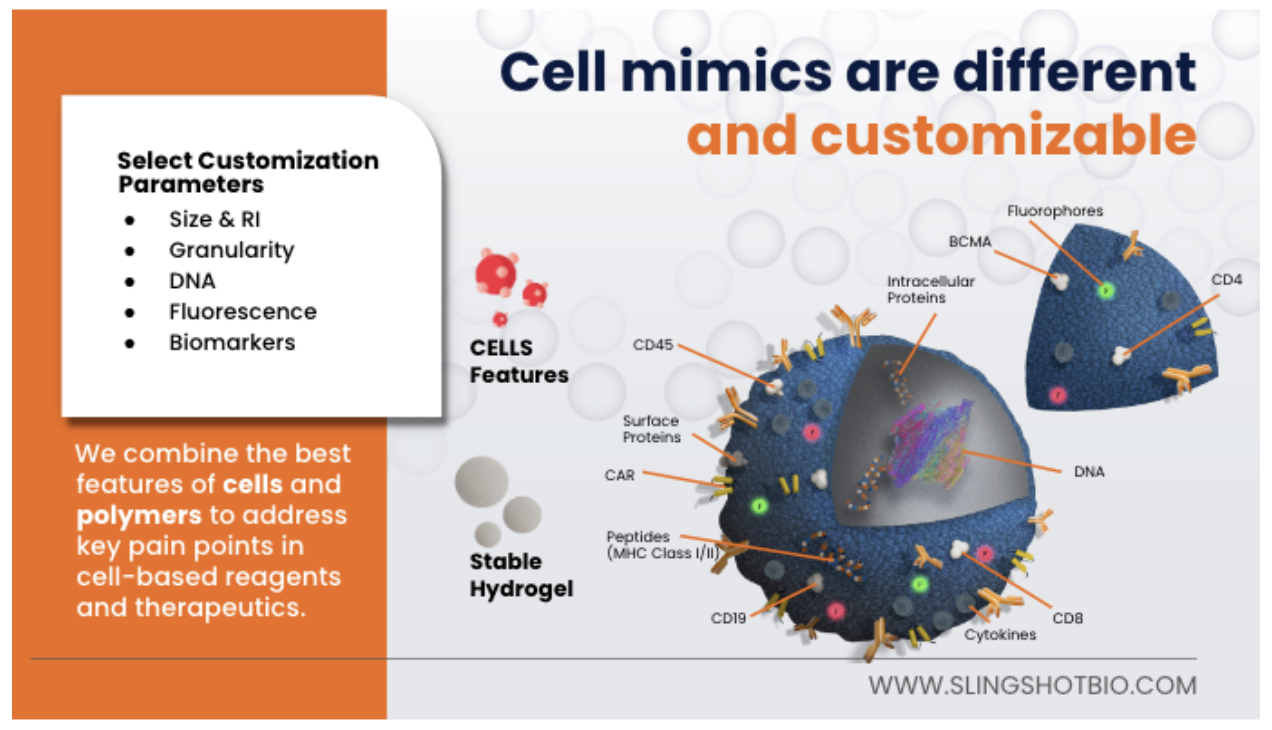
Single stained controls can also be used to compare your resolution with the multicolor sample. Fluorescence minus one (FMO) controls can be used to set gates accurately above the spread or to see which fluor in your panel may be impacting your spread if creating a SSM is not possible. Biomarker controls can be used to establish expression ranges but should also be used to monitor the performance of your flow assay over time. This can often be done through either commercially available blood based controls, or by freezing your cells. Both of these options come with their own sets of problems which can contribute to errors in your flow data, whether it be lot to lot variability, biohazardous material, or time consuming processes. So what do you do if you are working with a rare sample type or good positive controls are not available? This is where Slingshot Labs comes in. We have a curated selection of products for you to sample from or you can make your own control.
Conclusion
In summary, there are several areas in the flow cytometry workflow where errors can be introduced in your data. Following the best practices laid out in this post can help reduce or eliminate the errors in your data. At Slingshot we are “control freaks” who have made it our business to help others have the best flow data possible by offering non-biohazardous, consistent, reliable, and fully customizable cell mimics. Click the link below to learn more or reach out to sales@slingshotbio.com.
.png?width=50&name=Sarah_Kotanchiyev%20(1).png)